Introduction to Mode Sensors’ FDA-cleared Fluid Monitoring Wearable
In a recent development that underscores the growing impact of connected medical devices in healthcare, the U.S. Food and Drug Administration (FDA) cleared Mode Sensors' fluid monitoring wearable. This innovation represents a significant stride toward more continuous and accurate patient fluid monitoring, which is critical in managing a range of medical conditions, including heart failure, kidney disease, and postoperative care.
The Role of Connected Sensing in Modern Healthcare
Connected sensing technologies, such as Mode Sensors’ wearable, bring real-time, actionable data directly from patients to clinical teams. These devices can continuously track physiological parameters and transmit data securely to healthcare providers. This connectivity enables early detection of complications, timely interventions, and personalized care adjustments. The wearable's ability to provide continuous fluid status information exemplifies how sensors integrated with secure communication channels enhance the quality and timeliness of patient monitoring.
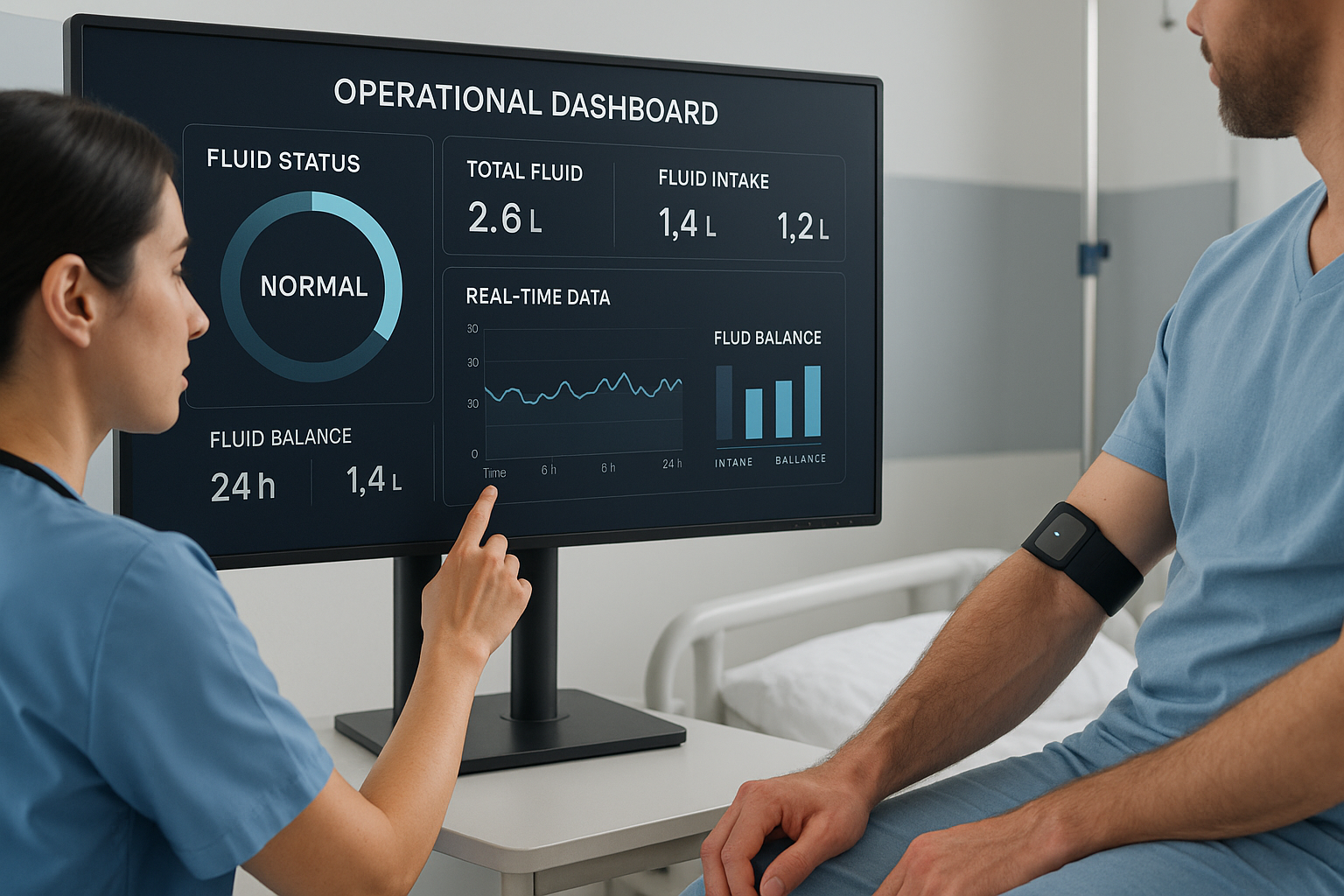
Operational Dashboards and Secure Data Flows for Improved Healthcare Delivery
Beyond data collection, the effective application of this information is crucial. Operational dashboards that aggregate data from connected devices enable healthcare professionals to visualize trends, prioritize patients requiring urgent attention, and optimize resource allocation. The secure flow of sensitive health data is essential to comply with regulatory standards such as HIPAA, maintaining patient privacy while enabling seamless information sharing across care teams. Integrating the data from wearables like Mode Sensors' device into these dashboards supports clinical decision-making and improves the reliability of care delivery.
Implications for Healthcare Systems and Providers
The FDA clearance of Mode Sensors' wearable signals broader trends in healthcare technology adoption. Medical centers and providers can leverage such technologies to transform patient monitoring from episodic to continuous, reducing hospital readmissions and improving outcomes. The scalability of secure connected sensing solutions facilitates remote patient management, easing pressure on healthcare facilities and enabling proactive interventions.
How Paw Partners Supports Implementation of Connected Medical Device Solutions
For healthcare organizations seeking to integrate advanced connected sensing devices like Mode Sensors’ fluid monitoring wearable into clinical workflows, Paw Partners offers engineering expertise in designing secure, reliable data infrastructures and user-friendly operational dashboards. Our experience across medical device connectivity, secure data integration, and system automation ensures that healthcare providers can realize the full benefits of emerging technologies with minimal disruption and maximum efficiency.